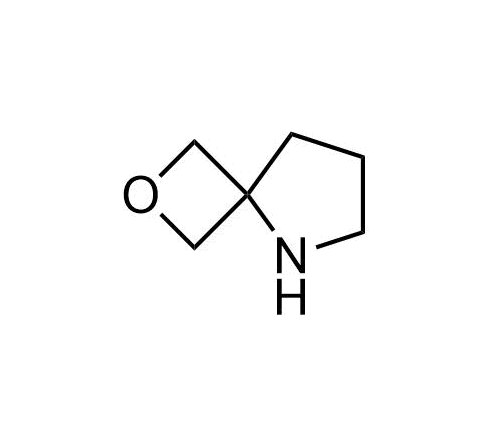
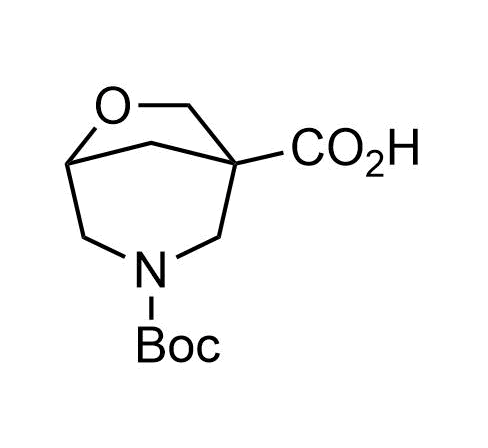
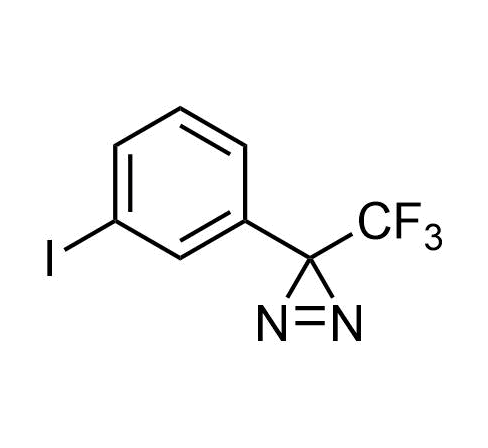
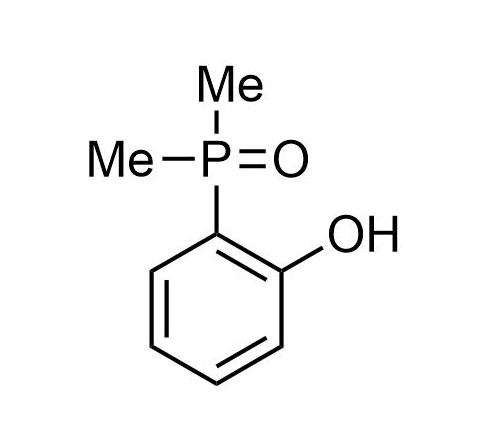
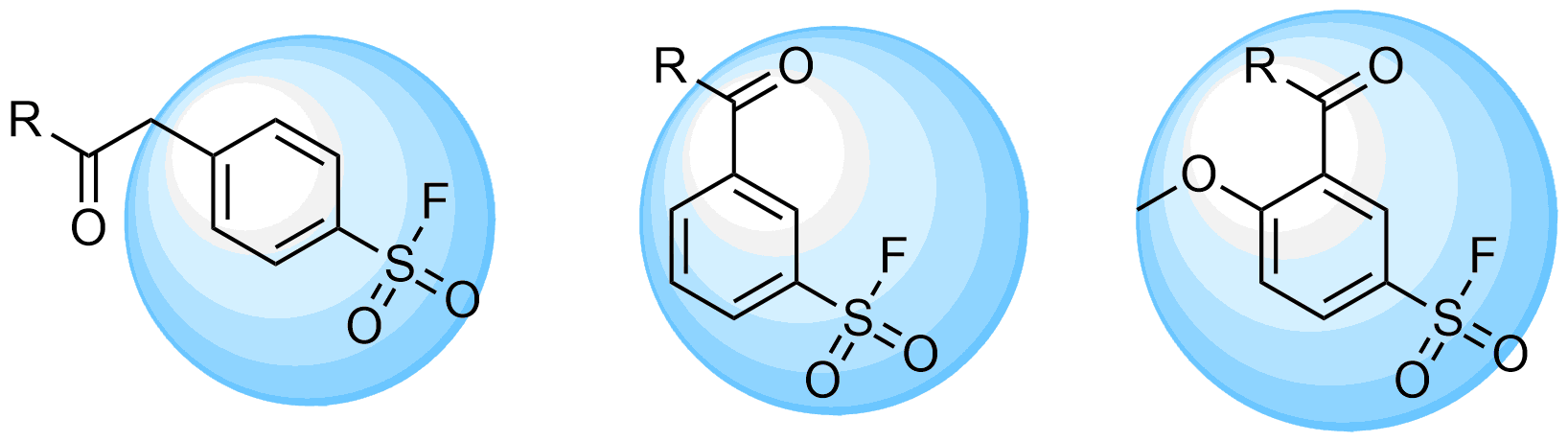
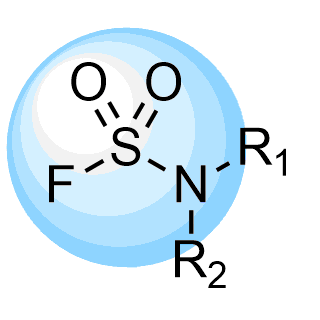
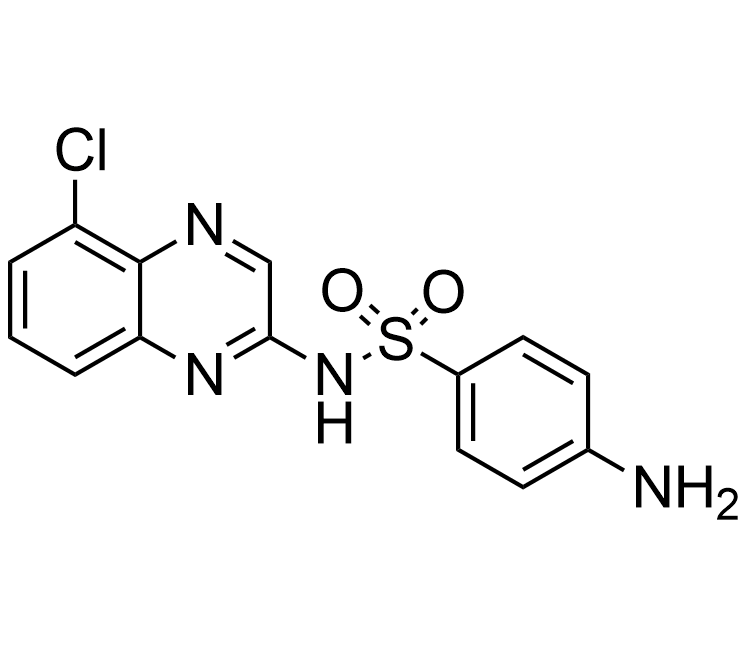
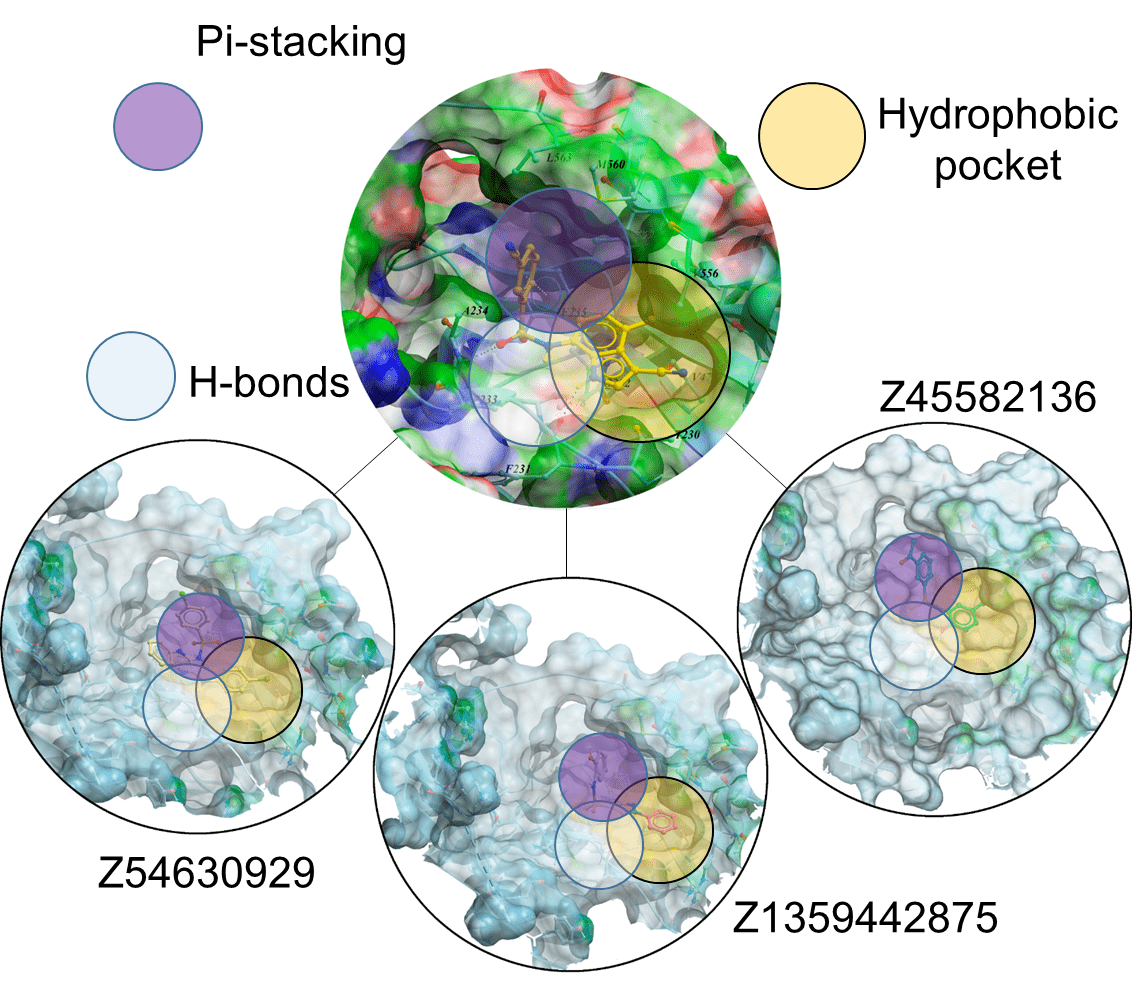
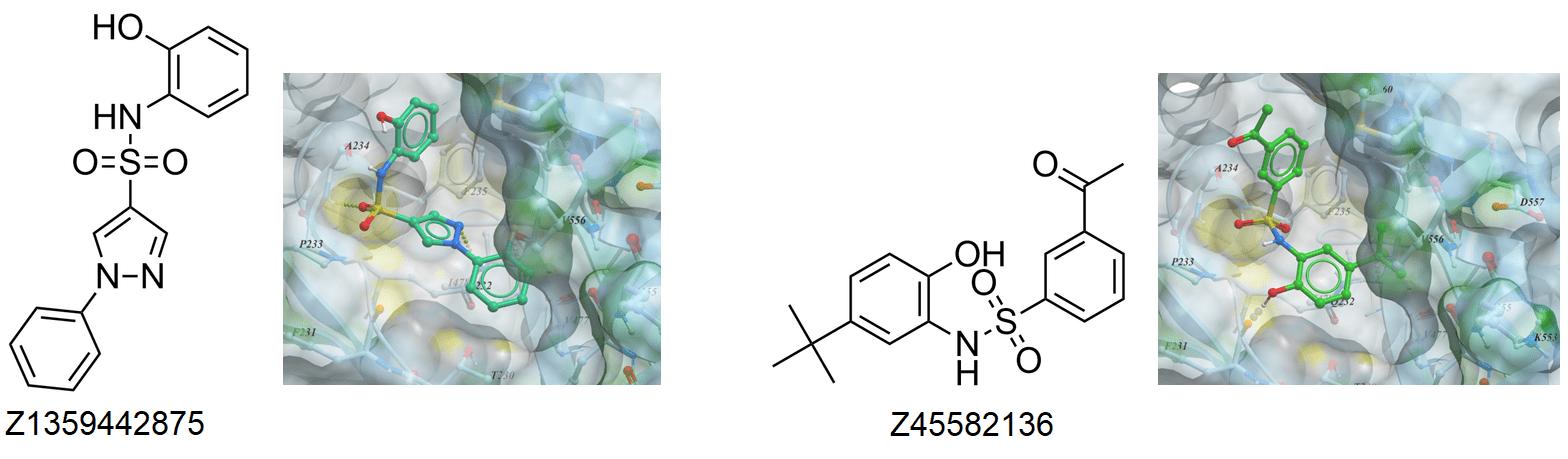
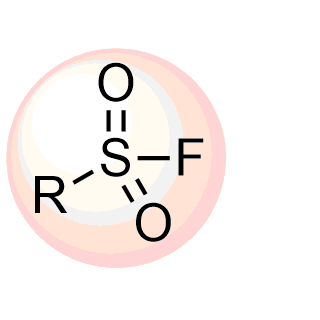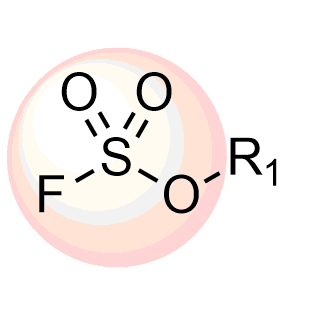Boronics Covalent Compounds
Over 5000 boron-containing compounds in stock
880 pre-plated compounds
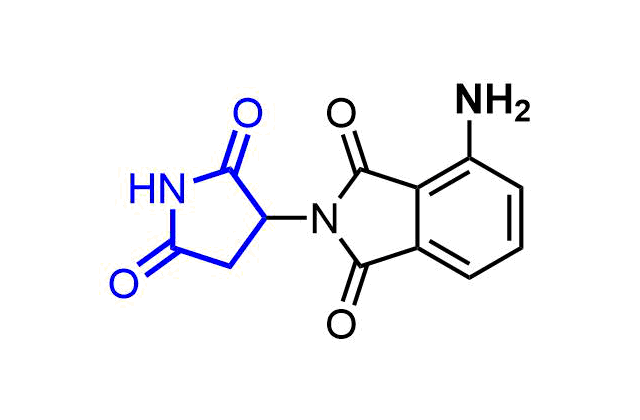
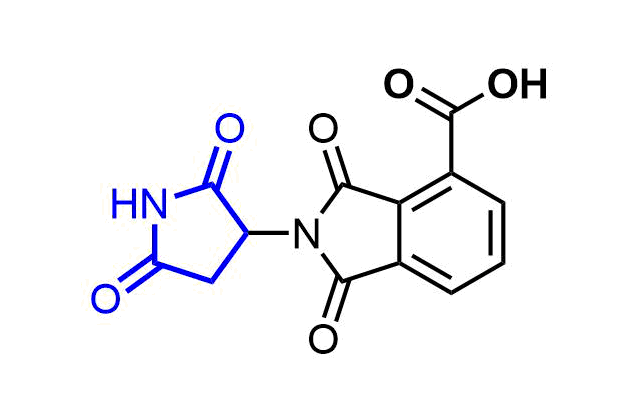
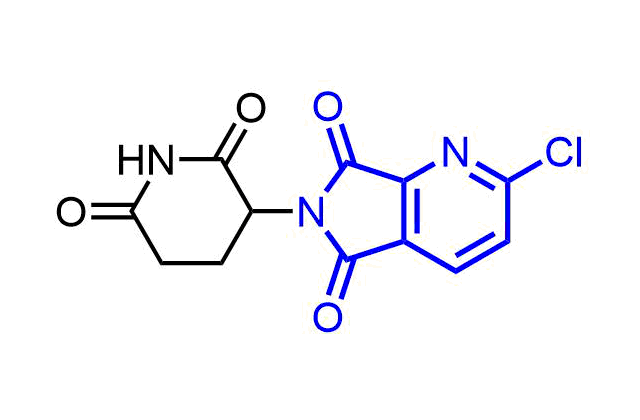
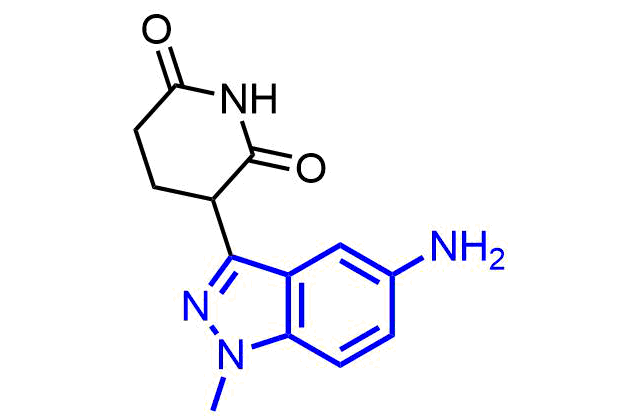
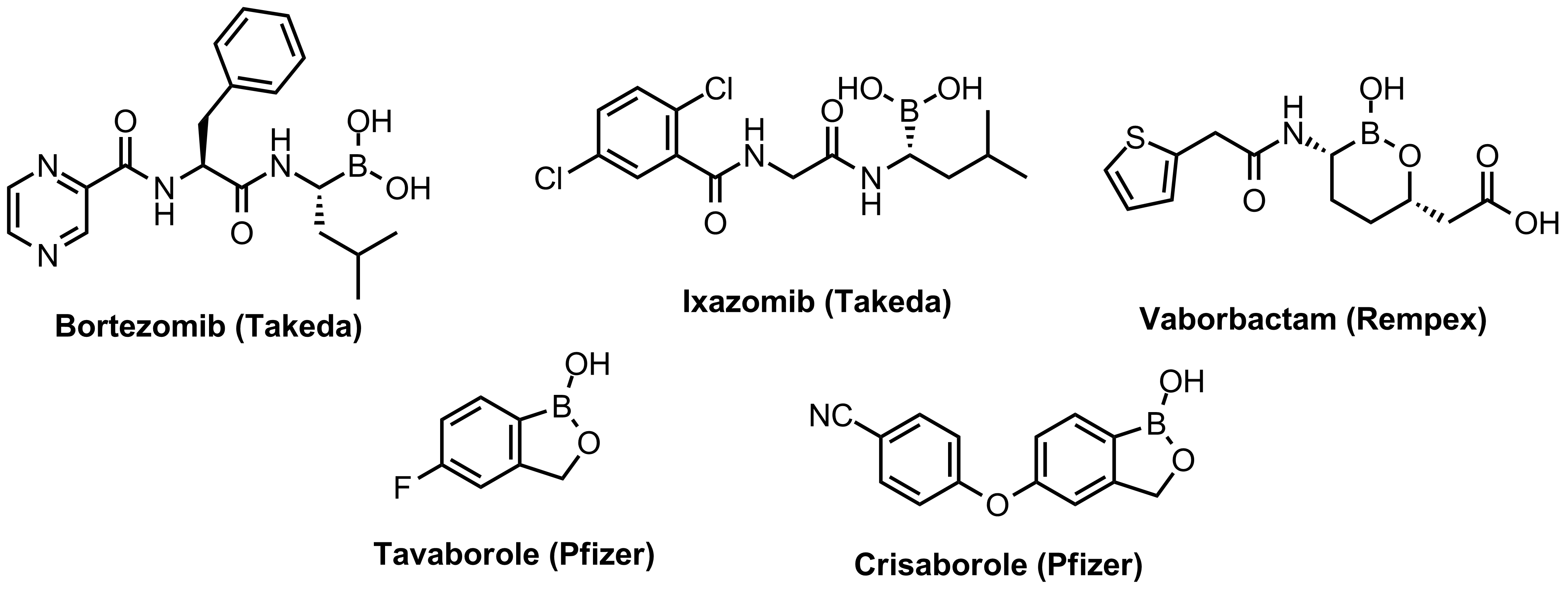
Boronics are mild and reversible covalent binders with proven efficacy and oral bioavailability. Bortezomib discovered as the first effective proteasome inhibitor validated the proteasome as a cancer target and revealed a novel approach in anticancer drug discovery. Further development of the next-generation proteasome inhibitor, the boronic ester prodrug Ixazomib citrate, has shown that boronic compounds can be administrated orally.
Other boron-containing drugs inhibit enzymes through formation of covalent adducts with catalytic nucleophilic residues like serine. The antifungal Tavaborole and phosphodiesterase-4 inhibitor Crisaborole, approved recently to treat psoriasis, contain benzoxaborole groups different from those reported before. More boronic compounds are in different pre-clinical and early clinical stages.
Approved Boronic Drugs
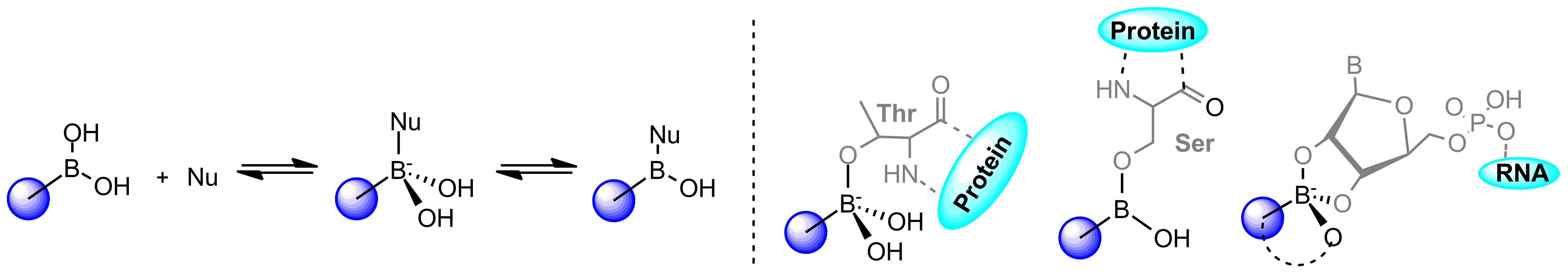
The high demand for boron-containing compounds is due to their unique electron structure. Although normally tri-coordinated, boronics readily adopts tetra-coordinated state upon exposure to nucleophiles and in such way can quickly form reversible adduct with nucleophilic residues such as serine, lysine, tyrosine, threonine, and cysteine thereby blocking the function of target proteins.
Boronic trigonal-tetragonal equilibrium, and covalent binding mode types
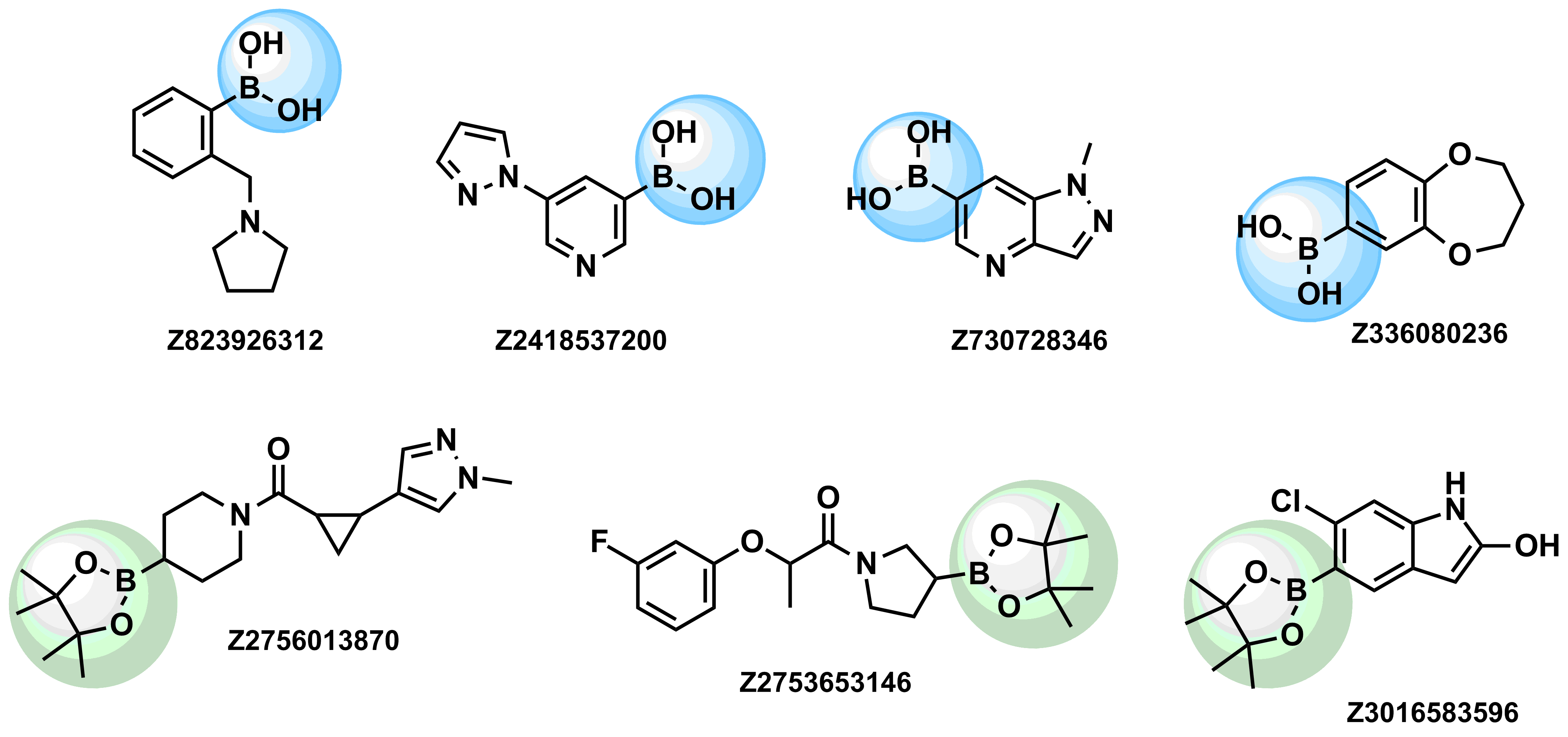
Enamine provides a wide range of boron compounds including boronic acids, and their more stable boronic ester analogues. In our library you could choose either aliphatic or (hetero)aromatic boronates.
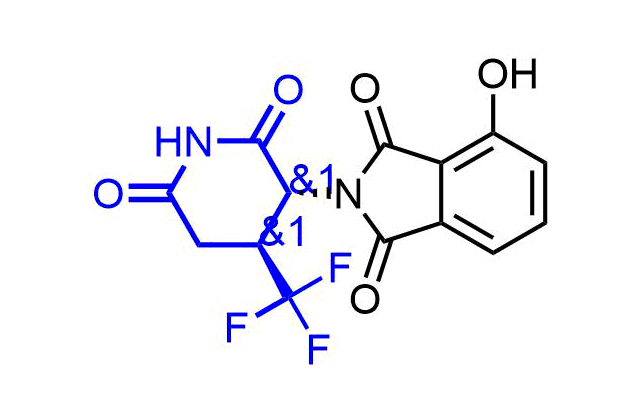
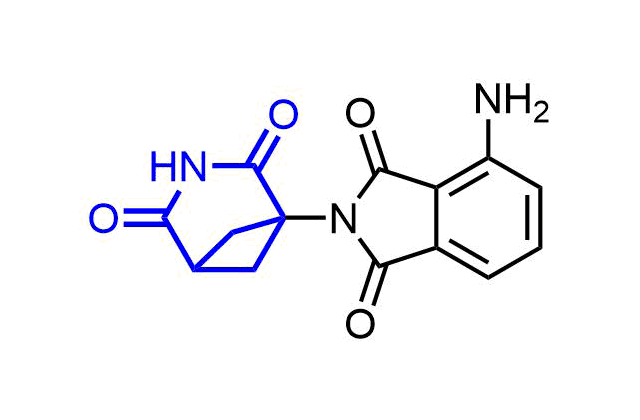
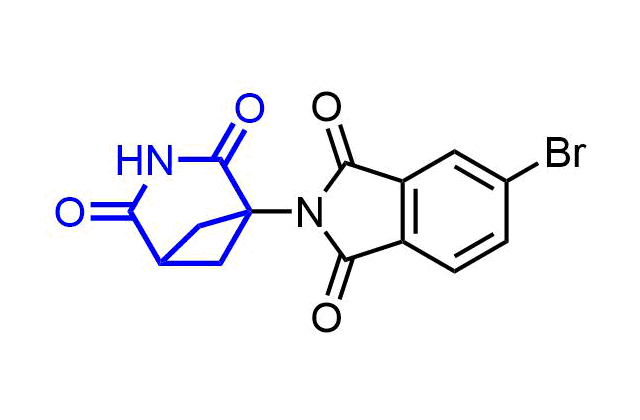
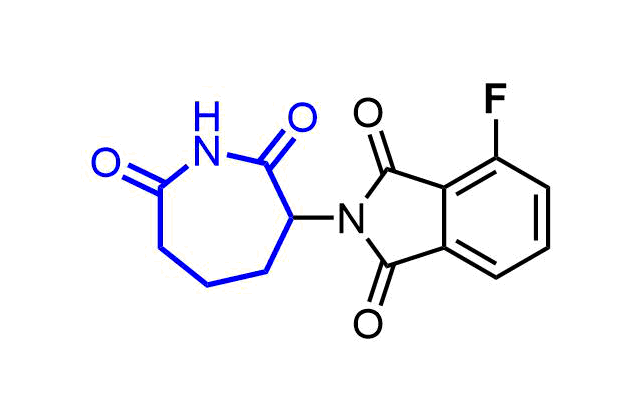
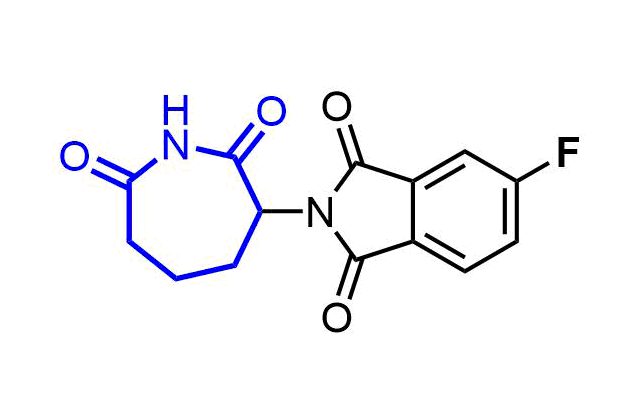
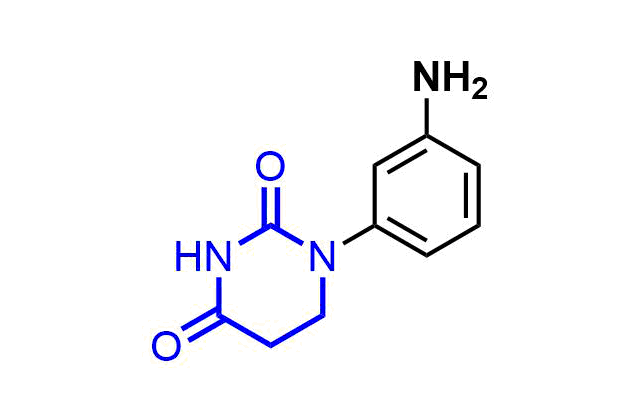
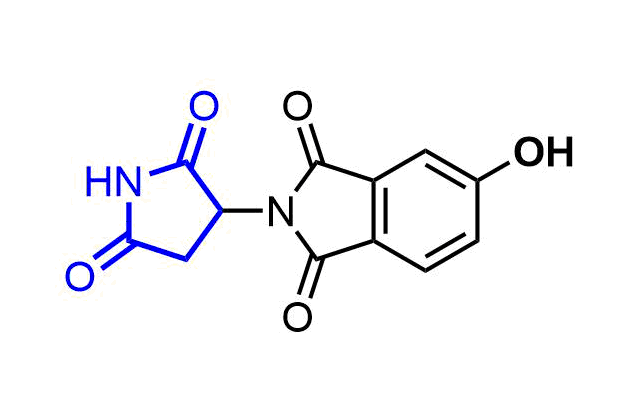
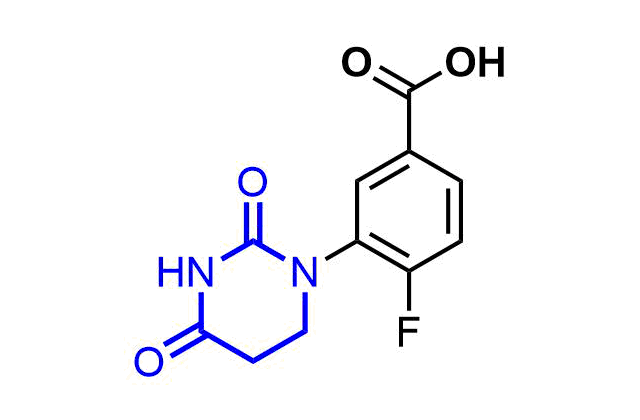
Examples of pre-plated Boronic Fragment
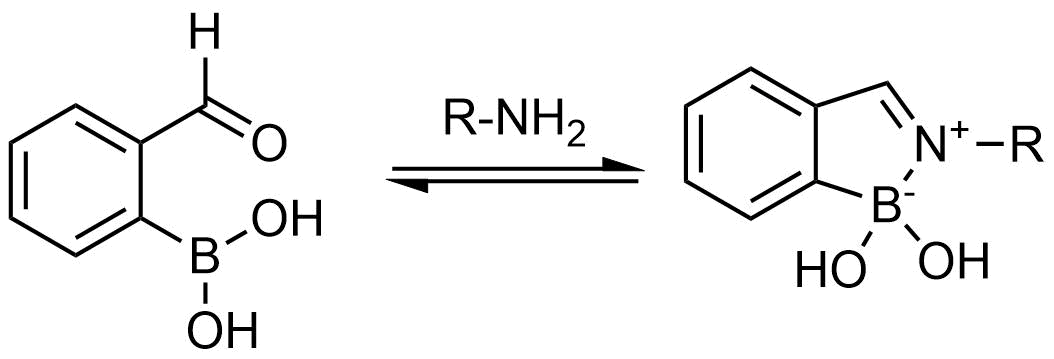
A growing number of studies are using aromatic α-formyl boronic acids as a selective warhead targeting lysine residue. Introduction of boronic acid residue into α-position of benzaldehydes (well-known covalent warhead) dramatically enhances stability of resulting Shiff bases by formation of 5-membered complex. The covalent adducts can be reversibly cleaved, that can be an advantage in case where irreversible off-target modification is observed.
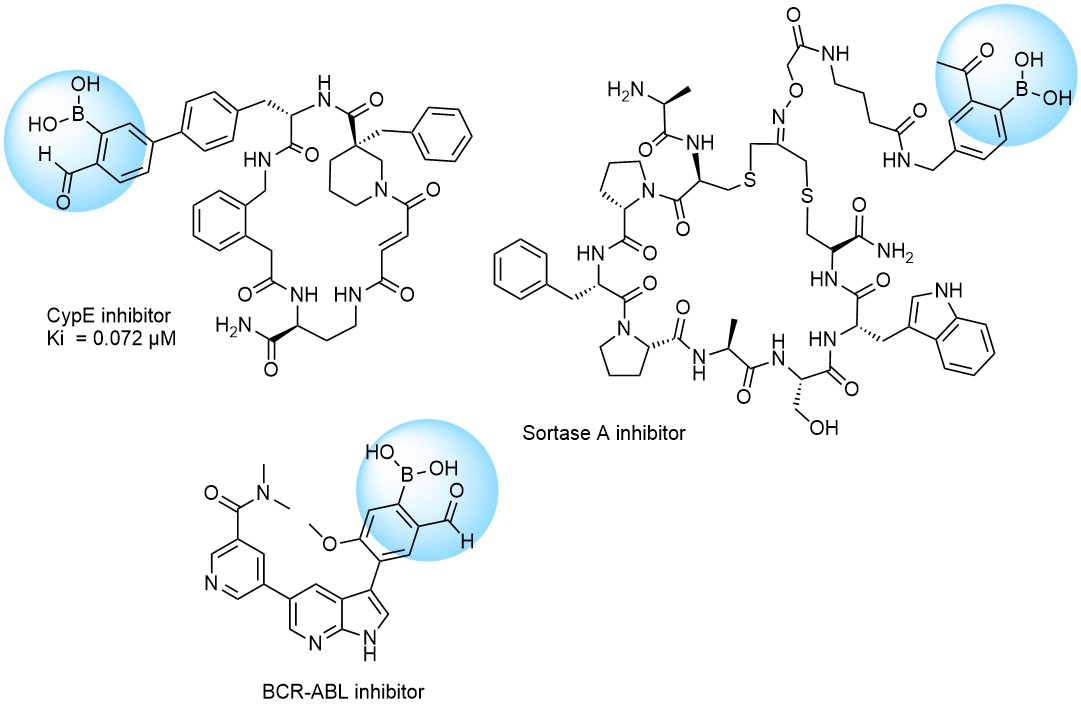
Recently research groups of Markus A. Seeliger & David R. Liu were developed subtype selective cyclophilin E reversible inhibitor with formyl boronic warhead covalently targeting lysine residue (Lys217)1. It is important that analogs with either the aldehyde or the boronic acid alone, respectively, showed reduced potency by 16-fold and 100-fold. Another example of α-carbonyl boronic warhead was developed into inhibitor of sortase A of S. aureus.
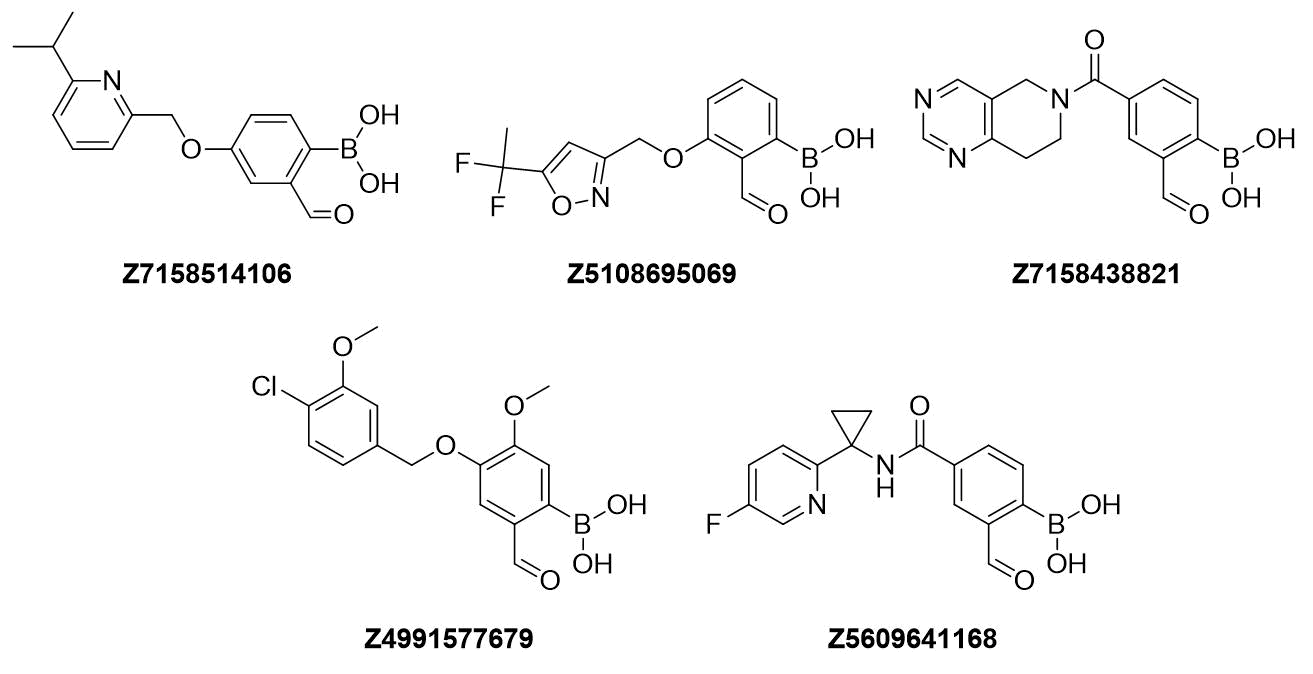
Examples of Formyl Boronates in pre-plated Covalent Screening Library
Typical Formats
Catalog No.
BCC-880-10-Y-10
Compounds
8803 plates
Amount
10 µL of 10 mM DMSO solutions
Format
384-well plates Greiner 781280, 320 compounds per plate, first two and last two columns empty
Price
Catalog No.
BCC-880-5-Y-10
Compounds
880 3 plates
Amount
5 µL of 10 mM DMSO solutions
Format
384-well plates Greiner 781280, 320 compounds per plate, first two and last two columns empty
Price